NOVUS Life Sciences standard medical-grade filaments include Bonlecule, PEEK, PMMA, PC, PLLA and PETG. They also provide customized, medical-grade filaments tailored for advanced healthcare applications, including sterile and non-sterile PEEK-HA (Hydroxyapatite), PLLA-HA, ABS, TPU, PCL, PCL-HA, PLGA, and PLGA-HA.
All the filaments have undergone rigorous testing and evaluation for biocompatibility in accordance with ISO 13485, ISO 10993, and USP Class VI standards. They are specifically designed to meet the stringent requirements for direct contact with skin and tissue for up to 24 hours, as well as, where applicable, indirect contact with blood.
For detailed specifications, material guidance, or custom formulation support, please contact our team for more information.


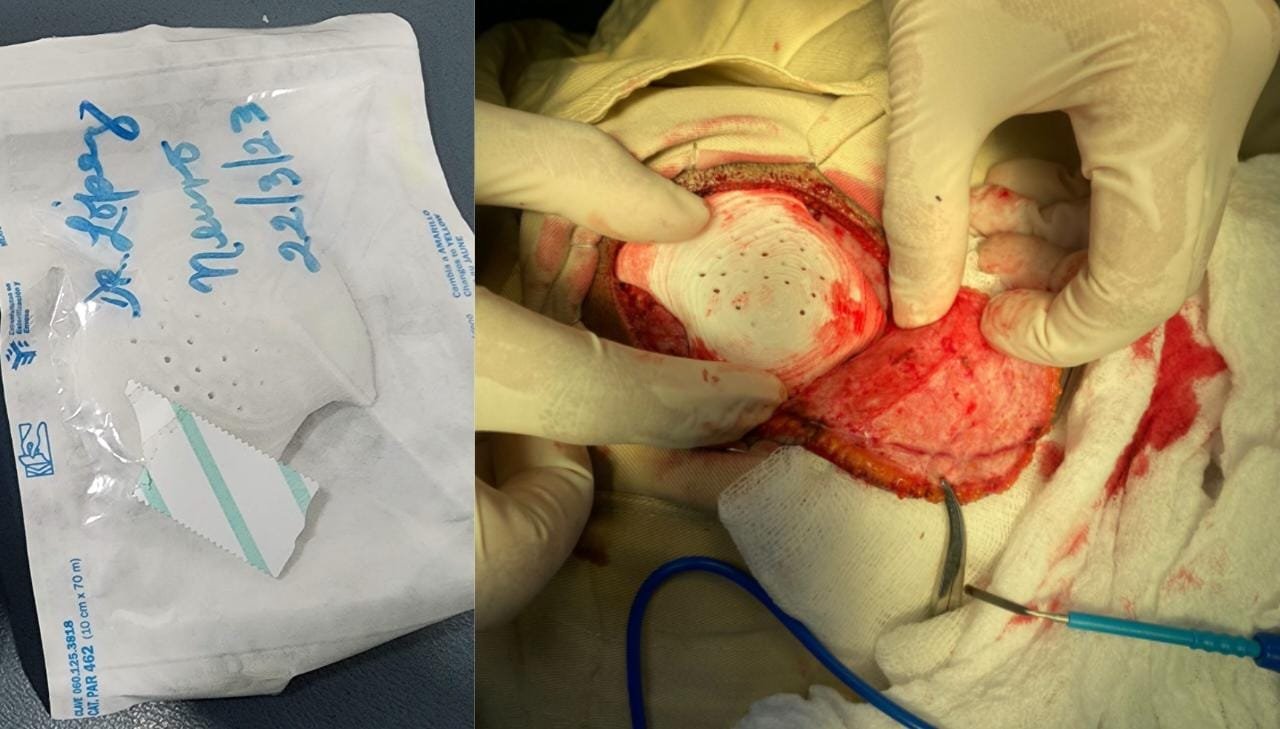
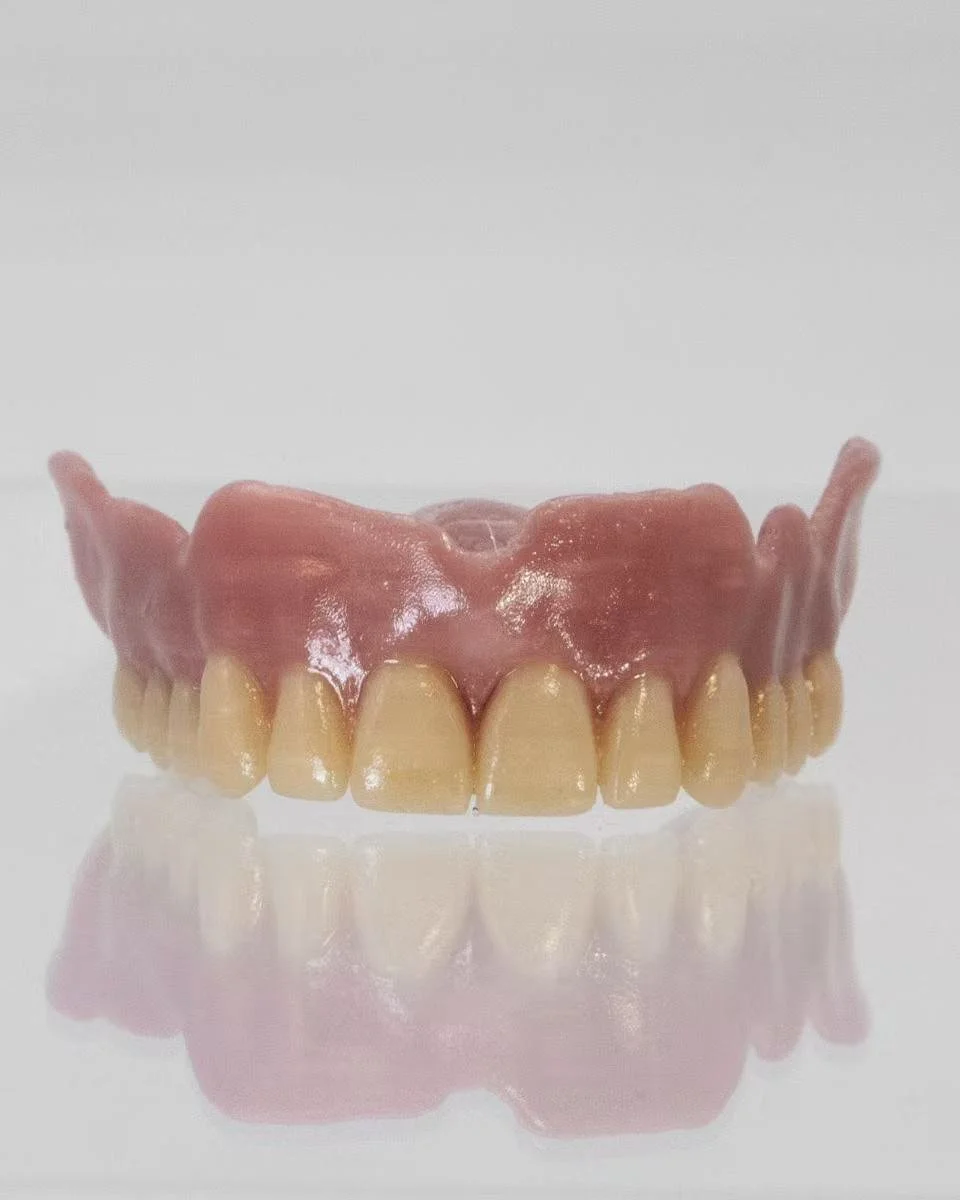
Binsei Inc., headquartered in Tokyo with a production facility in Yamanashi, Japan, is a manufacturer of high-performance materials. Leveraging many years of experience and expertise in compounding and precision extrusion, Binsei Inc. supply high-quality materials and products for medical and industrial applications.




